Stability Testing of Enzyme-linked Immunosorbent Assay Kit
The stability of ELISA kit may refer to the ability to maintain the quality and efficiency of the kit during the shelf life defined by the Manufacturer. According to different experimental purpose and condition, Stability tests can be divided into accelerated stability tests and long-term stability test.
The accelerated stability tests,is normally performed at 37℃ to accelerate the chemical or physical reaction of biological reagents in the kit. And then the stability of the kit is determined. Since many unexpected factors may exist during the transportation or storage, the stability of the kit under such conditions can be estimated by the accelerated stability test. And it can be also used to predict the shelf life of the kits which are stored at normal condition.
Keep the kits at 37℃ for 24 hours and 72hours, and then arrange recovery test, linearity test and precision test to validate the kit.
The long-term stability test, also called the stability test within the period of validity, is performed at normal storage condition to validate the efficiency of kits stored at normal condition. It is usually used to reflect the stability of the kit directly since the result of the long-term stability test is the final basis to determine the shelf life and storage condition. Arrange the recovery test, linearity test and precision test of the kits within 30 days before the expiration date and within 30 days after the expiration date to validate the kit.
In order to make sure the efficiency of the kit, all the kits are passed the Accelerated stability test and long long-term stability test before releasing them into the market. Take SEA056Ra(L141030739) for example,the data is shown below.
1. Accelerated stability test
Recovery
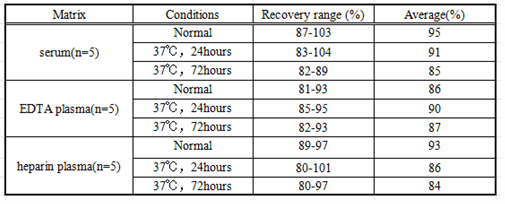
Matrices listed below were spiked with certain level of recombinant IL10 and the recovery rates were calculated by comparing the measured value to the expected amount of IL10 in samples.
Table1: the result of recovery test for the kits stored in normal condition, kept at 37℃ for 24 hours and kept at 37℃ for 72 hours.
Linearity
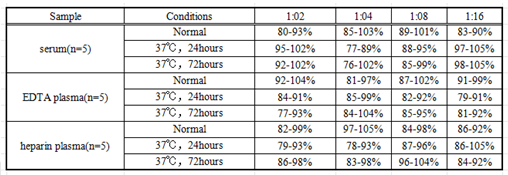
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of IL10 and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.(Linearity:70-130%)
Table2: the result of linearity test for the kits stored in normal condition, kept at 37℃ for 24 hours and kept at 37℃ for 72 hours.
Precision
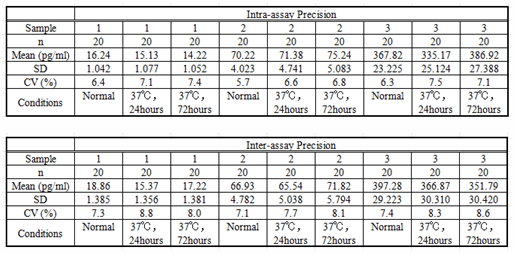
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level IL10 were tested 20 times on one plate, respectively.
2. Stability test within the period of Validity
Recovery
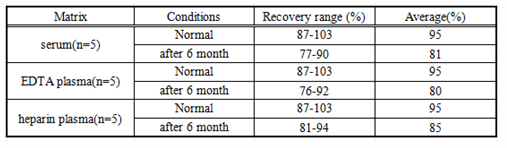
Matrices listed below were spiked with certain level of recombinant IL10 and the recovery rates were calculated by comparing the measured value to the expected amount of IL10 in samples. (Recovery: 70-130%)
Table4: the result of recovery test for the kits within 30 days before the expiration date and within 30 days after the expiration date.
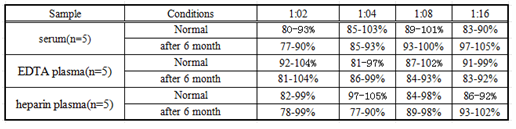
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of IL10 and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.(Linearity:70-130%)
Table5: the result of linearity test for the kits within 30 days before the expiration date and within 30 days after the expiration date.
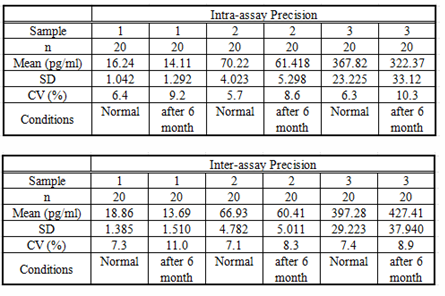
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level IL10 were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level IL10 were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Table6: the result of precision test for the kits within 30 days before the expiration date and within 30 days after the expiration date.
For more information,please visit http://www.cloud-clone.us/。
