Immunohistochemistry (IHC) is used for identification and localization of the target antigen/antibody by observing the staining results of sections. During the process of sectioning, it is essential to maintain the integrity of antigens. Dissolution and denaturation of antigen should be avoided during the process of staining, washing and embedding. Meanwhile, it should be eliminated that antigens diffuse to near by.
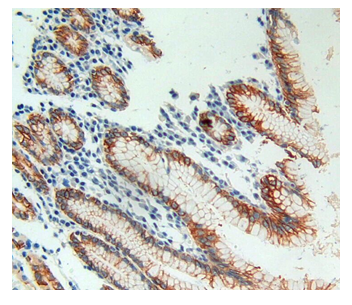
Figure1 IHC image cells or tissue spaces.
Tissue processing is important for obtaining good result of IHC. Living tissue, various body fluid, punctate and cells can be used for IHC analysis. Different process methods can be used according to various specimens. For living tissue, Paraffin tissue sections and frozen tissue sections is usually recommended. Body fluid, punctate and cells are processed by smear method. For bacterial colony or lesion tissues from dead organism, printing slices can be made by pressing fresh section on the slide. Herein we will introduce the common tissue processing technology for living tissue-frozen tissue section and paraffin tissue section.
1.Frozen tissue section
1) Method
①Materials. Select freshly isolated tissue, cut into small pieces of approximately 2mm thickness.
②Quick freezing. Place the tissue piece into a clean box, apply OCT embedding compound to completely cover the tissue. Drop the box into liquid nitrogen and remain there for 10-20s for fast freezing. Store at −80℃ for long-term storage.
③Sectioning. Bring the freezing microtome to the cryostat, cut 5- to 10-µm-thick sections. The thickness of the section is based on the type of tissue. If the tissue contains experimental cells that are particularly large, thin sections should be cut. For tissue containing plenty of fiber but few cells, thick sections should be cut. The freezing degree will depends on the tissue type. For example, when cut non-fixed brain tissue, liver tissue and lymph node, the temperature should not be too low, -10 to -15 °C is proper. For tissues including thyroid, spleen, kidney and muscle, the temperature can be adjusted to be -15 to -10 °C. For tissue containing lipid, the temperature should be -25 °C. If there is plenty of lipid in the tissue, -30 °Cis appropriate.-20s for fast freezing. Store at −80°C for long-term storage.
2) Suitable specimens: unstable antigen
3) Advantages: simple operation, suitable for unstable antigen.
4) Disadvantage: The tissues’ structure may be not clear
2.Paraffin tissue section
1) Method
①Materials. Choose suitable area, cut into 2-3 mm -thick sections.
②Fixation. Wash the tissue with normal saline. Immerse the tissue fragment in 10% neutral buffered formalin for 30-50 min.
③Dehydration. Tissue is dehydrated through a graded ethanol series.
50% ethanol(0.5h)————>70% ethanol(0.5h)————>80% ethanol(0.5h)————>95% ethanol(0.5h) ————>100% ethanol(0.5h) ————>100% ethanol
④Hyalinization. Immerse the tissue fragment in three-level hyalinization solutions after dehydration. The tissue become transparent after that.
1/2 Xylene+1/2 ethanol(2h)————>100% Xylene(1.5h)————>100% Xylene(1.5h)
⑤Waxing. After hyalinization, immerse the tissue in 1/2 xylene and 1/2 ethanol, then keep it in 40°C incubator for 40 min. Apply paraffin I for 30 min, paraffin II for 40 min to exclude the hyalinization solution. Waxing should be carried out in an incubator and the incubation time depends on the size of the tissue.
⑥Embedding. Adjust the temperature to 60 °C, exchange wax for three 3 times, incubating 1-2 h each time.
⑦Sectioning. Put the fixed paraffin blocks on the microtome. Cut into 4-10um-thickness sections and observe it under a microscope.
⑧Sticking. Put gelatin subbing solution on a clear slide, add a drop of distilled water and paste the sections on the slide. Transfer the slide onto a slide warmer at 35°C, then remove liquid using absorbent paper and dry slide on the warmer.
2) Advantage: a clear preservation of fine of tissue will be obtained.
3) Disadvantage: antigen in tissue will partially lose activity during the preparation of Paraffin tissue section.

