New discovery on the mechanism of sepsis
Sepsis, regarded as one of the oldest and most elusive syndromes in medicine, is a common condition in which tissues and organs are severely damaged due to a dysregulated host responses. Epidemiological data have shown that sepsis is a serious public health issue and may contribute to high morbidity and mortality worldwide, and this trend is increasing annually.
New discovery on the mechanism of sepsis
Currently, the classical treatment of sepsis relies on appropriate antibiotics and steroid hormones to eradicate the source of infection as soon as possible, which is crucial for favorable outcome in sepsis. However, the precise temporal benefits of antibiotics may be controversial due to the unfavorable consequences of antibiotic treatment, such as drug resistance and adverse reactions. Furthermore, numerous clinical trials on immunomodulation in sepsis have failed to improve patient outcome. Therefore, a deeper understanding of the disease mechanism of sepsis is needed to develop better strategies to improve the prognosis of patients with sepsis.
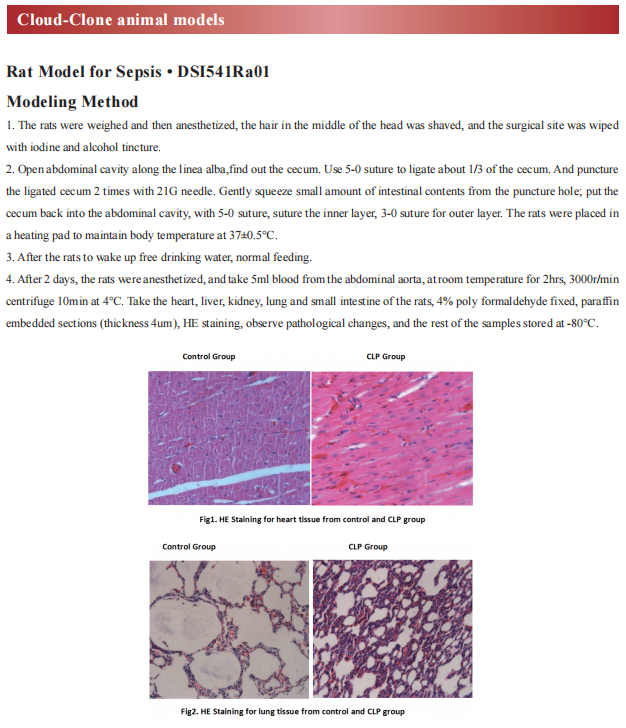
1. APOL1 risk variants drive endothelial cell defects that exacerbate sepsis
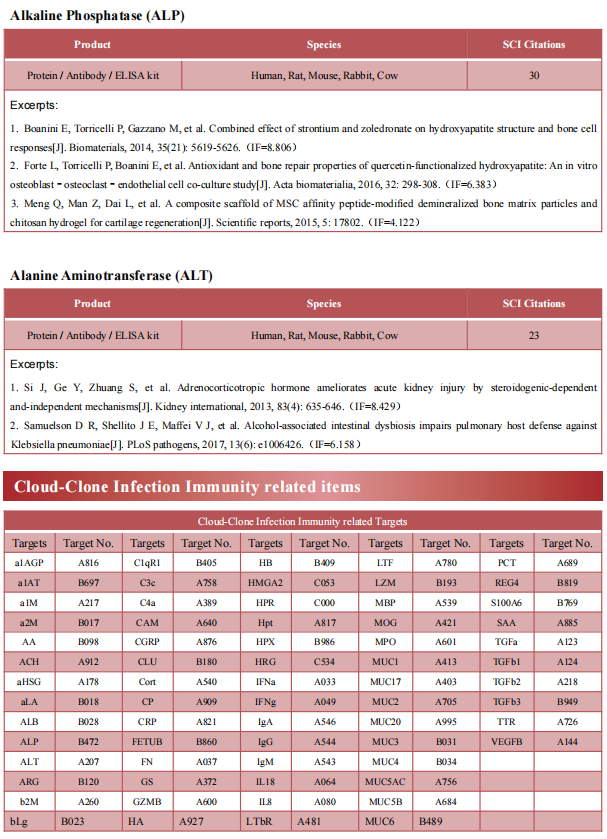
Katalin Susztak, Department of Medicine, Perelman School of Medicine, University of Pennsylvania, and hia team found the incidence and severity of sepsis is higher among individuals of African versus European ancestry.[1] Further analysis revealed that that genetic risk variants (RVs) in the trypanolytic factor apolipoprotein L1 (APOL1), present only in individuals of African ancestry, were associated with increased sepsis incidence and severity. Analysis of mice with endothelial-specific expression of RV APOL1 and in vitro studies demonstrated that RV APOL1 interfered with mitophagy, leading to cytosolic release of mitochondrial DNA and activation of the inflammasome (NLRP3) and the cytosolic nucleotide sensing pathways (STING) (Fig.1). Genetic deletion or pharmacological inhibition of NLRP3 and STING protected mice from RV APOL1-induced permeability defects and proinflammatory endothelial changes in sepsis. The work identifies the role of endothelial RV APOL1 in sepsis among Black individuals, highlights the key role of mitochondrial dysfunction and activation of the inflammasome and cGAS and STING, and emphasizes critical therapeutic targets for intervention.
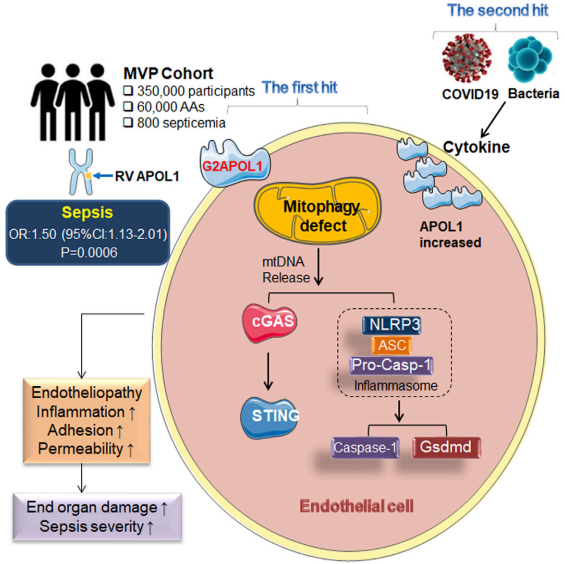
Fig.1 RV APOL1 leads to endothelial cell defects, activation of NLRP3 inflammosome and STING exacerbate sepsis
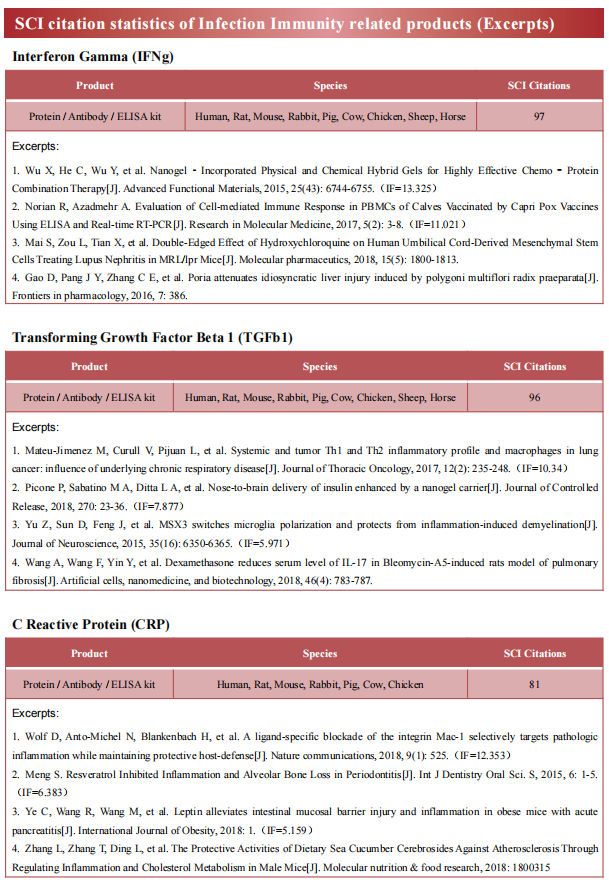
2. Platelet MHC Class I Mediates CD8+ T Cell Suppression During Sepsis
In sepsis, platelet-leukocyte interactions are increased, and have been associated with adverse clinical events, including increased platelet-T cell interactions. Matthew T. Rondina, University of Utah Molecular Medicine Program, USA, and his team systemically evaluated platelet antigen internalization and presentation through major histocompatibility complex class I (MHC-I) and their effects on antigen specific CD8+ T cells in sepsis in vivo and ex vivo.[2] The expression of platelet MHC-I is significantly increased in human and murine platelets during sepsis and in human megakaryocytes stimulated with agonists generated systemically during sepsis (e.g., IFN-g and LPS). Upregulation of platelet MHC-I during sepsis increases antigen cross-presentation and interactions with CD8+ T cells in an antigen-specific manner. Using a platelet lineage specific MHC-I deficient mouse strain (B2mf/f--Pf4Cre), we demonstrate that platelet MHC-I regulates antigen-specific CD8+ T cell proliferation in vitro, as well as the number and functional responses of CD8+ T cells in vivo during sepsis (Fig.2). Loss of platelet MHC-I reduced sepsis-associated mortality in mice in an antigen specific setting. The results from this study indicate that through platelet MHC-I, platelets internalize, process, and present antigens to CD8+ T cells. Through this mechanism, increased MHC-I on platelets during sepsis results in suppressed CD8+ T cell number, proliferation, and functional responses.
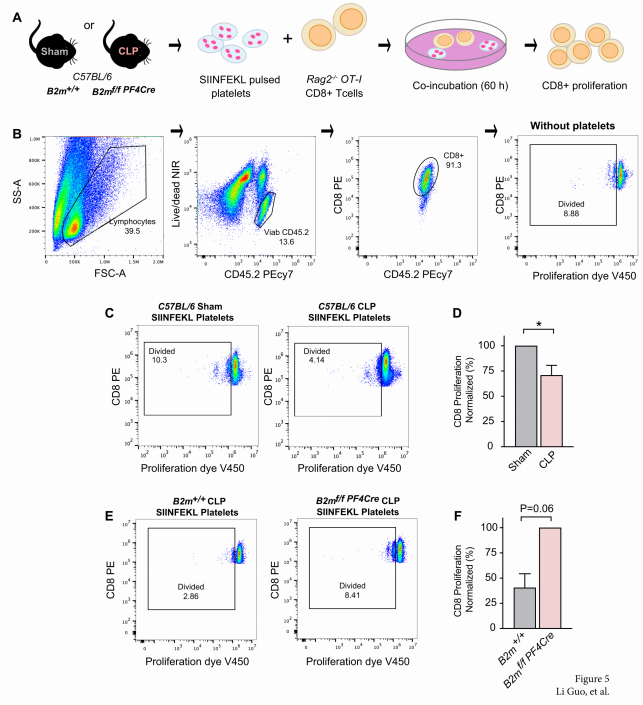
Fig.2 Platelets from septic mice directly induce antigen specific CD8+ T cell suppression in vitro
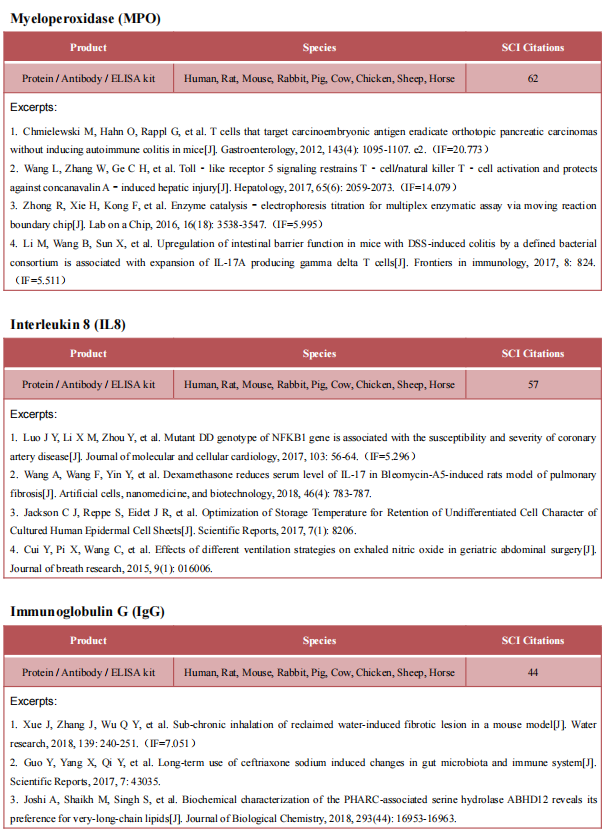
3. Sepsis expands a CD39+ plasmablast population that promotes immunosuppression via adenosine-mediated inhibition of macrophage antimicrobial activity
Sepsis results in elevated adenosine in circulation. Extracellular adenosine triggers immunosuppressive signaling via the A2a receptor (A2aR). Sepsis survivors develop persistent immunosuppression with increased risk of recurrent infections. Jose´ Carlos Alves-Filho, University of Sa˜ o Paulo, Brazil, and his team utilized the cecal ligation and puncture (CLP) model of sepsis and subsequent infection to assess the role of adenosine in post-sepsis immune suppression.[3] A2aR-deficient mice showed improved resistance to post-sepsis infections. Sepsis expanded a subset of CD39hi B cells and elevated extracellular adenosine, which was absent in mice lacking CD39-expressing B cells. Sepsis-surviving B cell-deficient mice were more resistant to secondary infections. Mechanistically, metabolic reprogramming of septic B cells increased production of ATP, which was converted into adenosine by CD39 on plasmablasts. Adenosine signaling via A2aR impaired macrophage bactericidal activity and enhanced interleukin-10 production (Fig.3). The data therefore suggest that adenosine can be a potential therapeutic target for persistent immunosuppression in sepsis survivors.
Fig.3 Sepsis expands a CD39+ plasmablast population that promotes immunosuppression via adenosine-mediated inhibition of macrophage antimicrobial activity
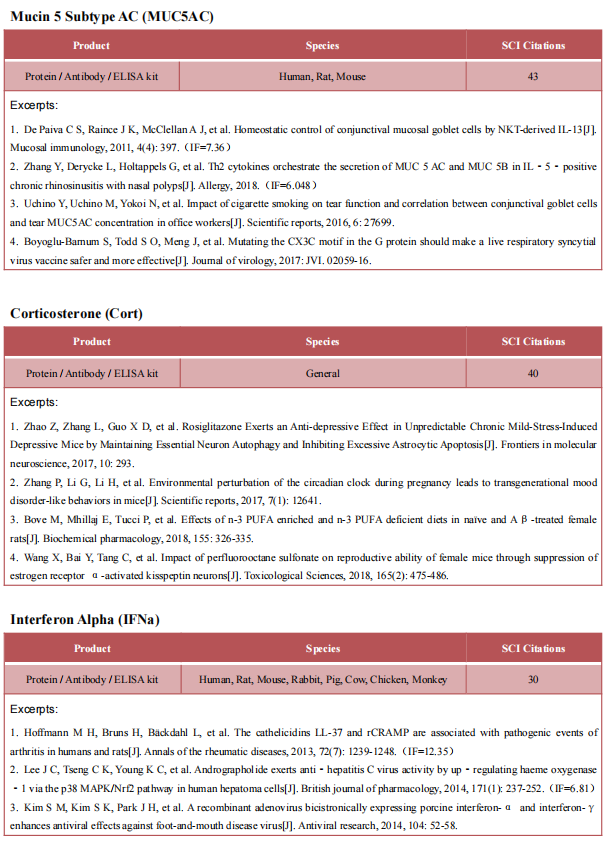
4. Uncoupling of IL-6 signaling and LC3-associated phagocytosis drives immunoparalysis during sepsis
Immune deactivation of phagocytes is a central event in the pathogenesis of sepsis. Georgios Chamilos, School of Medicine, University of Crete, Greece, and his team identified a master regulatory role of IL-6 signaling on LC3-associated phagocytosis (LAP) and reveal that uncoupling of these two processes during sepsis induces immunoparalysis in monocytes/macrophages.[4] In sepsis, loss of IL-6 signaling specifically abrogates microtubule-mediated trafficking of ERK, leading to defective activation of LAP and impaired killing of bacterial and fungal pathogens by monocytes/macrophages, which can be selectively restored by IL-6 supplementation. The findings link IL-6 signaling with LAP, with an important role in the development of immunoparalysis of sepsis (Fig.4), which can be explored as a therapeutic target.
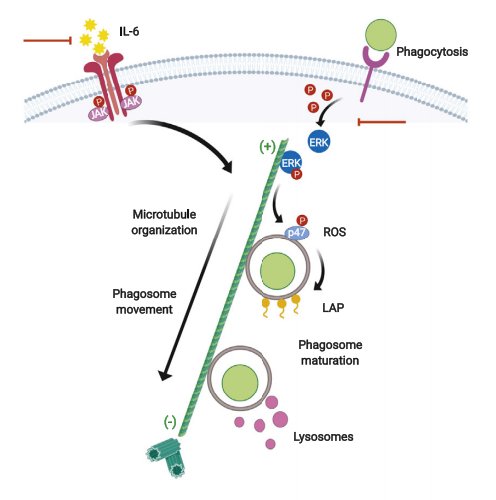
Fig.4 Uncoupling of IL-6 signaling and LC3-associated phagocytosis drives immunoparalysis during sepsis
References
[1]Wu J, Ma Z, Raman A, et al. APOL1 risk variants in individuals of African genetic ancestry drive endothelial cell defects that exacerbate sepsis [J]. Immunity. 2021, 54(11):2632-2649.e6. (IF=31.745)
[2]Guo L, Shen S, Rowley JW, et al. Platelet MHC class I mediates CD8+ T-cell suppression during sepsis [J]. Blood. 2021, 138(5):401-416. (IF=22.113)
[3]Nascimento DC, Viacava PR, Ferreira RG, et al. Sepsis expands a CD39+ plasmablast population that promotes immunosuppression via adenosine-mediated inhibition of macrophage antimicrobial activity [J]. Immunity. 2021, 54(9):2024-2041.e8. (IF=31.745)
[4]Akoumianaki T, Vaporidi K, Diamantaki E, et al. Uncoupling of IL-6 signaling and LC3-associated phagocytosis drives immunoparalysis during sepsis [J]. Cell Host Microbe. 2021;29(8):1277-1293.e6. (IF=21.023)
Cloud-Clone not only provide animal disease models of sepsis, but also have various infection-immune-related protein indicator detection products and the above mentioned APOL1, NLRP3, STING, MHC-I, A2aR, IL-6 and other protein related products, which can help the general scientific research workers to conduct sepsis related research.