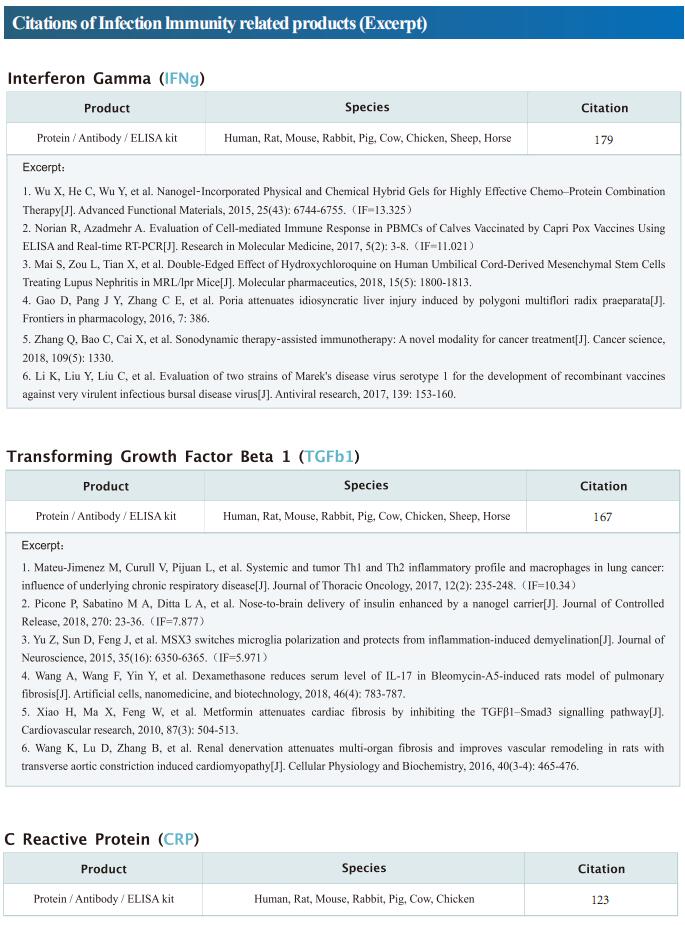
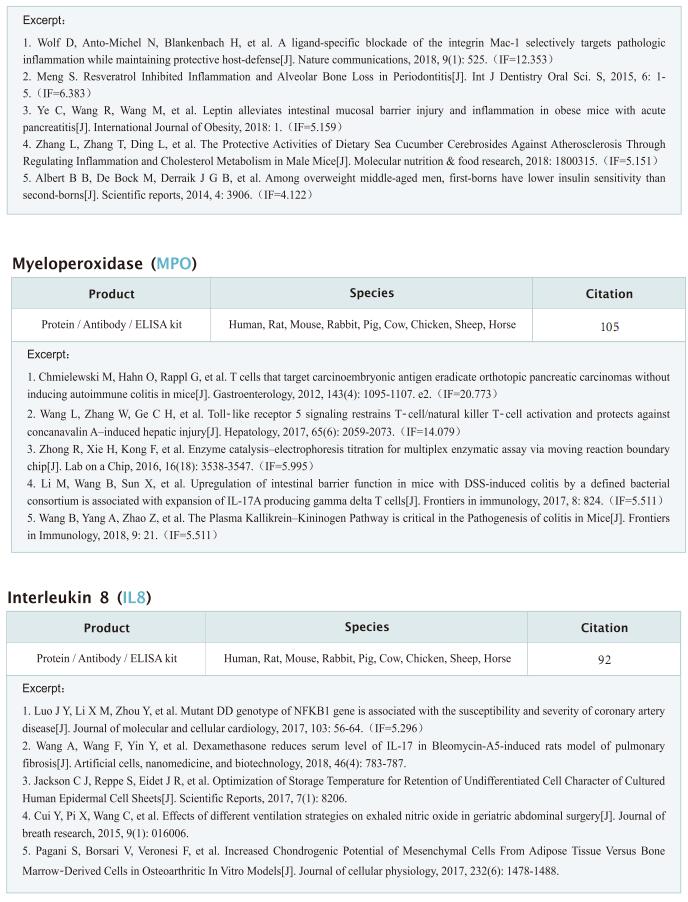
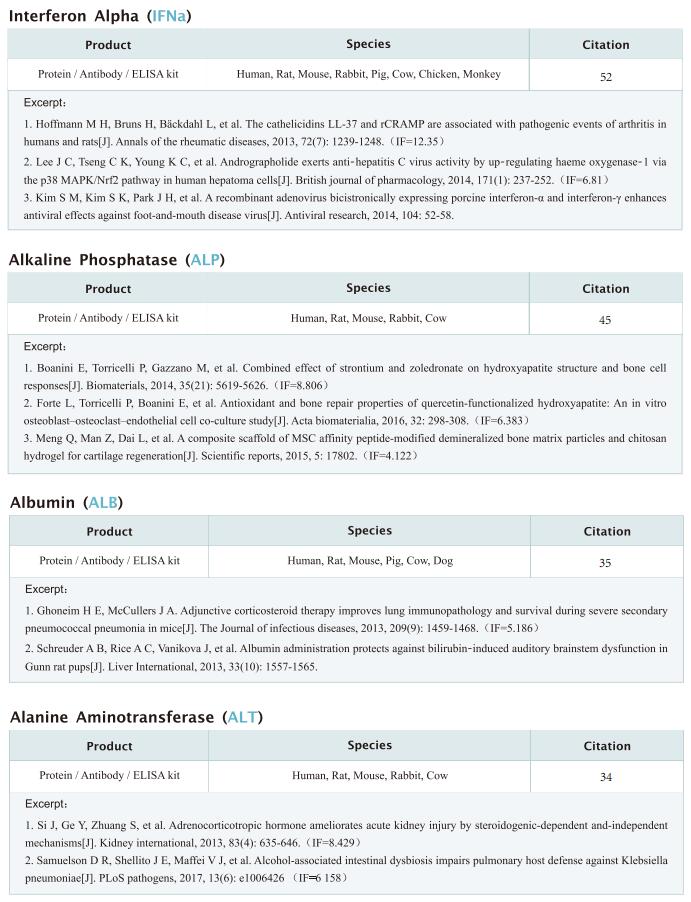
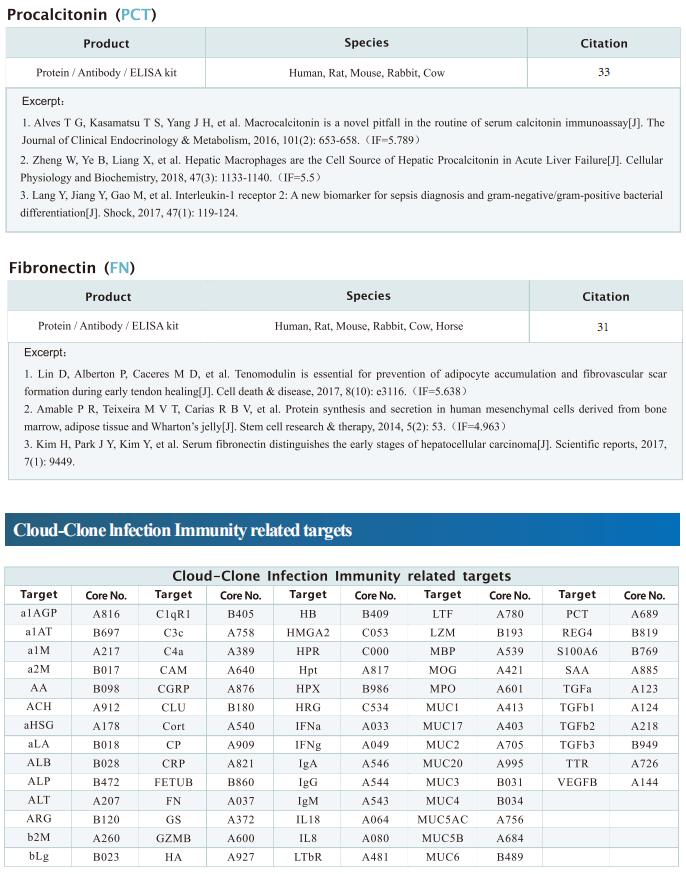
Targeting mitochondria regulates inflammation and immunity
Remodeling of mitochondrial dynamics and mitochondrial morphology plays a pivotal role in the maintenance of mitochondrial homeostasis in response to pathogenic attacks or stress stimuli. In addition to their role in metabolism and energy production, mitochondria participate in diverse biological functions, including innate immune responses driven by macrophages in response to infections or inflammatory stimuli. Innate immune responses involving sensing of microbes and generation of antimicrobial effectors are the primary defense in response to pathogenic attacks and damage signals. Accumulating evidence suggests that multilayered molecular crosstalk between innate immune signaling and mitochondrial dynamics regulates the outcomes of pathogenic infections. Recently, several articles have reported the role of mitochondria in the crosstalk between innate immunity, immune metabolism and inflammation, which may provide help for the treatment and prevention of related diseases.
1. An LKB1–mitochondria axis controls TH17 effector function
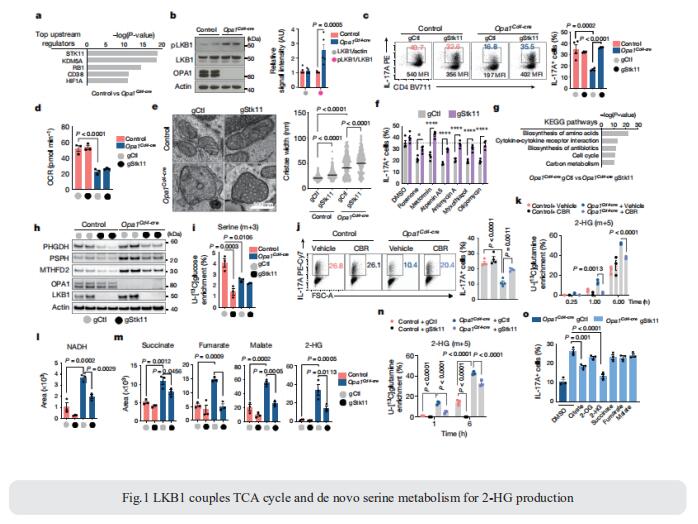
Erika L. Pearce, Max Planck Institute for Immunobiology and Epigenetics, Germany, and his team showed that unlike other CD4+ T cell subsets, T helper 17 (TH17) cells have fused mitochondria with tight cristae[1]. Their multi-omics approach revealed that the serine/threonine kinase liver-associated kinase B1 (LKB1) couples mitochondrial function to cytokine expression in TH17 cells by regulating tricarboxylic acid (TCA) cycle metabolism and transcriptional remodelling. Mitochondrial membrane disruption activates LKB1, which restrains IL-17 expression. LKB1 deletion restores IL-17 expression in TH17 cells with disrupted mitochondrial membranes, rectifying aberrant TCA cycle glutamine flux, balancing NADH/NAD+ and preventing 2-hydroxyglutarate production from the promiscuous activity of the serine biosynthesis enzyme phosphoglycerate dehydrogenase (Fig.1). These data suggest that an LKB1–mitochondrial axis sustains T cell metabolic adaptations to distinct inflammatory and tissue conditions, and illustrate metabolic and mitochondrial vulnerabilities in TH17 cells that might be therapeutically exploited to control autoimmune disease.
2. Augmenting NK cell-based immunotherapy by targeting mitochondrial apoptosis
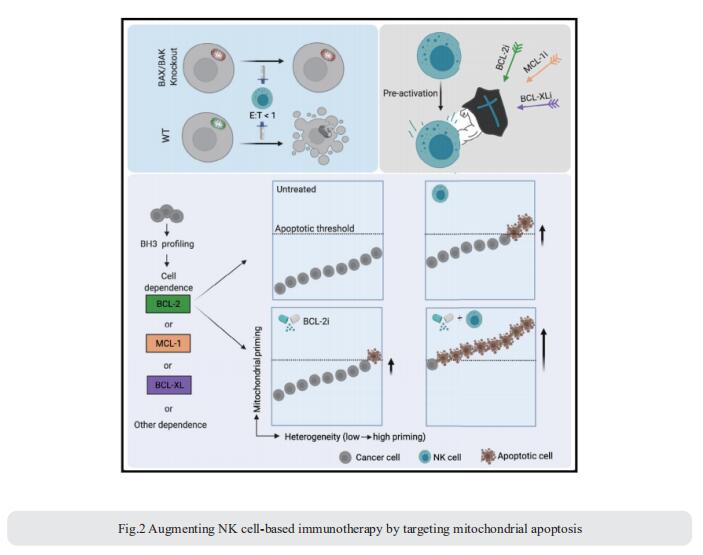
Interest in harnessing natural killer (NK) cells for cancer immunotherapy is rapidly growing. However, efficacy of NK cell-based immunotherapy remains limited in most trials. Anthony Letai, Department of Medical Oncology, Dana-Farber Cancer Institute, USA, and his team found that mitochondrial apoptosis (mtApoptosis) pathway is essential for efficient NK killing, especially at physiologically relevant effector-to-target ratios[2]. Furthermore, NK cells can prime cancer cells for mtApoptosis and mitochondrial priming status affects cancer-cell susceptibility to NK-mediated killing. Combining BH3 mimetics with NK cells synergistically kills cancer cells in vitro and suppresses tumor growth in vivo (Fig.2). They herein report a rational and precision strategy to augment NK-based immunotherapy, which may be adaptable to T cell-based immunotherapies as well.
3. Mitochondrial electron transport chain is necessary for NLRP3 inflammasome activation
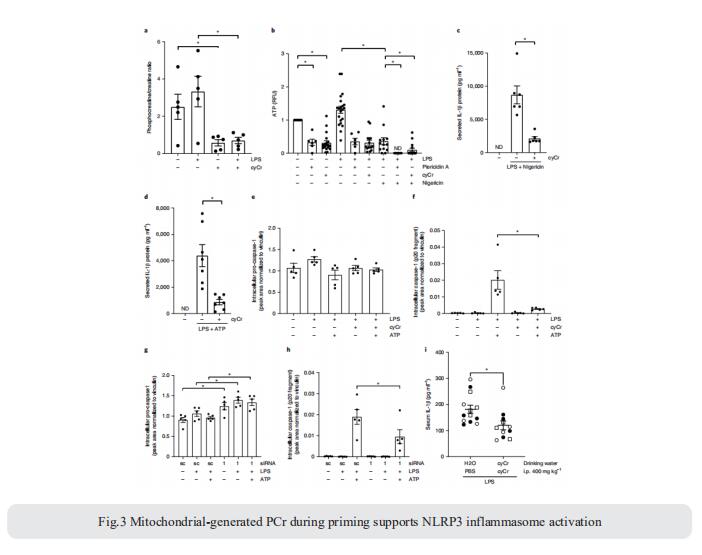
The NLRP3 inflammasome is linked to sterile and pathogen-dependent inflammation, and its dysregulation underlies many chronic diseases. Mitochondria have been implicated as regulators of the NLRP3 inflammasome through several mechanisms including generation of mitochondrial reactive oxygen species (ROS). Navdeep S. Chandel, Department of Medicine, Northwestern University Feinberg School of Medicine, USA, reported that mitochondrial electron transport chain (ETC) complex I, II, III and V inhibitors all prevent NLRP3 inflammasome activation[3]. Ectopic expression of Saccharomyces cerevisiae NADH dehydrogenase (NDI1) or Ciona intestinalis alternative oxidase, which can complement the functional loss of mitochondrial complex I or III, respectively, without generation of ROS, rescued NLRP3 inflammasome activation in the absence of endogenous mitochondrial complex I or complex III function. Metabolomics revealed phosphocreatine (PCr), which can sustain ATP levels, as a common metabolite that is diminished by mitochondrial ETC inhibitors. PCr depletion decreased ATP levels and NLRP3 inflammasome activation (Fig.3). Thus, the mitochondrial ETC sustains NLRP3 inflammasome activation through PCr-dependent generation of ATP, but via a ROS-independent mechanism.
4. Mitochondrial ROS promotes susceptibility to infection via gasdermin D-mediated necroptosis
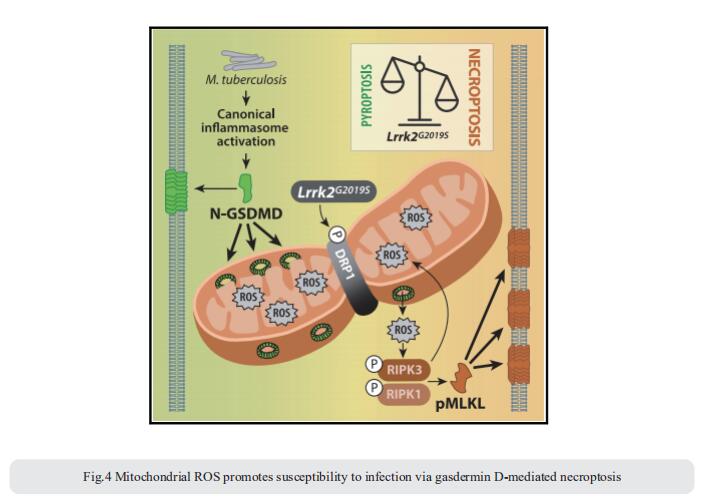
Although mutations in mitochondrial-associated genes are linked to inflammation and susceptibility to infection, their mechanistic contributions to immune outcomes remain ill-defined. Robert O. Watson, Department of Microbial Pathogenesis and Immunology, Texas A&M Health, College of Medicine, USA, and his team discovered that the disease-associated gain-of-function allele Lrrk2G2019S (leucine-rich repeat kinase 2) perturbs mitochondrial homeostasis and reprograms cell death pathways in macrophages[4]. When the inflammasome is activated in Lrrk2G2019S macrophages, elevated mitochondrial ROS (mtROS) directs association of the pore-forming protein gasdermin D (GSDMD) to mitochondrial membranes. Mitochondrial GSDMD pore formation then releases mtROS, promoting a switch to RIPK1/RIPK3/MLKL-dependent necroptosis. Consistent with enhanced necroptosis, infection of Lrrk2G2019S mice with Mycobacterium tuberculosis elicits hyperinflammation and severe immunopathology. Their findings suggested a pivotal role for GSDMD as an executer of multiple cell death pathways and demonstrate that mitochondrial dysfunction can direct immune outcomes via cell death modality switching. This work provides insights into how LRRK2 mutations manifest or exacerbate human diseases and identifies GSDMD-dependent necroptosis as a potential target to limit Lrrk2G2019S-mediated immunopathology.
References
[1]Baixauli F, Piletic K, Puleston DJ, et al. An LKB1-mitochondria axis controls TH17 effector function [J]. Nature. 2022;610(7932):555-561. (IF=69.504)
[2]Pan R, Ryan J, Pan D, Wucherpfennig KW, Letai A. Augmenting NK cell-based immunotherapy by targeting mitochondrial apoptosis [J]. Cell. 2022;185(9):1521-1538.e18. (IF=66.850)
[3]Billingham LK, Stoolman JS, Vasan K, et al. Mitochondrial electron transport chain is necessary for NLRP3 inflammasome activation [J]. Nat Immunol. 2022;23(5):692-704. (IF=31.250)
[4]Weindel CG, Martinez EL, Zhao X, et al. Mitochondrial ROS promotes susceptibility to infection via gasdermin D-mediated necroptosis [J]. Cell. 2022;185(17):3214-3231.e23. (IF=66.850)
Cloud-Clone not only provides a variety of commonly used infection immune detection indicators related products, including PCT, CRP, CGRP, IL-6, IL-8, IL-10, HMGB1, SAA, IFNα, etc., but also provides detection products related to apoptosis and pyroptosis signaling pathways, which can help the majority of scientific researchers to conduct mitochondrial and immune related studies.