Packages (Simulation)

Reagent Preparation
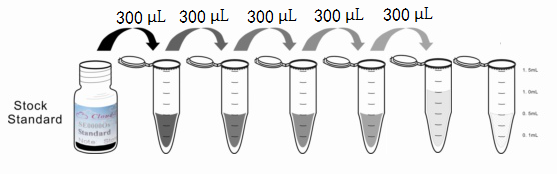
Image (I)
Image (II)
Certificate


Multiplex Assay Kit for Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay)
CSF2; GMCSF; Sargramostim; Molgramostin; Granulocyte-Macrophage Colony Stimulating Factor
(Note: Up to 8-plex in one testing reaction)
- Product No.LMA045Po
- Organism SpeciesSus scrofa; Porcine (Pig) Same name, Different species.
- Sample TypeSerum, plasma, tissue homogenates, cell lysates, cell culture supernates and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3.5h
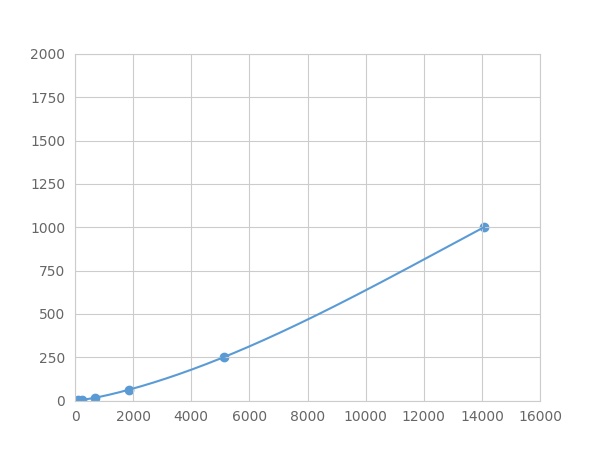
- Detection Range0.98-1000pg/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 0.327 pg/mL.
- DownloadInstruction Manual
- UOM 8Plex 7Plex 6Plex 5Plex 4Plex 3Plex 2Plex1Plex
- FOB
US$ 472
US$ 490
US$ 517
US$ 553
US$ 590
US$ 644
US$ 726
US$ 907
Add to Price Calculator
Result
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay).
No significant cross-reactivity or interference between Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) and the recovery rates were calculated by comparing the measured value to the expected amount of Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 78-105 | 102 |
| EDTA plasma(n=5) | 89-98 | 94 |
| heparin plasma(n=5) | 91-105 | 98 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 85-101% | 87-101% | 88-96% | 80-101% |
| EDTA plasma(n=5) | 80-92% | 98-105% | 89-99% | 80-101% |
| heparin plasma(n=5) | 95-104% | 94-102% | 95-102% | 81-97% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| 96-well plate | 1 | Plate sealer for 96 wells | 4 |
| Pre-Mixed Standard | 2 | Standard Diluent | 1×20mL |
| Pre-Mixed Magnetic beads (22#:GM-CSF) | 1 | Analysis buffer | 1×20mL |
| Pre-Mixed Detection Reagent A | 1×120μL | Assay Diluent A | 1×12mL |
| Detection Reagent B (PE-SA) | 1×120μL | Assay Diluent B | 1×12mL |
| Sheath Fluid | 1×10mL | Wash Buffer (30 × concentrate) | 1×20mL |
| Instruction manual | 1 |
Assay procedure summary
1. Preparation of standards, reagents and samples before the experiment;
2. Add 100μL standard or sample to each well,
add 10μL magnetic beads, and incubate 90min at 37°C on shaker;
3. Remove liquid on magnetic frame, add 100μL prepared Detection Reagent A. Incubate 60min at 37°C on shaker;
4. Wash plate on magnetic frame for three times;
5. Add 100μL prepared Detection Reagent B, and incubate 30 min at 37°C on shaker;
6. Wash plate on magnetic frame for three times;
7. Add 100μL sheath solution, swirl for 2 minutes, read on the machine.
GIVEAWAYS
INCREMENT SERVICES
| Magazine | Citations |
| Journal of Toxicology and Environmental Health | Effects of 900-MHz Microwave Radiation on γ-Ray-Induced Damage to Mouse Hematopoietic System PubMed: 20391130 |
| 5 | Safety and efficacy of the peptide-based therapeutic vaccine for HIV-1, Vacc-4×: a phase 2 randomised, double-blind, placebo-controlled trial Pubmed:24525316 |
| Archives of Virology | Genetic and immunogenicity analysis of porcine circovirus type 2 strains isolated in central China Pubmed:29305646 |
| BioMed Research International | Activated Macrophages of Monocytic Origin Predominantly Express Proinflammatory Cytokine Genes, Whereas Kupffer Cells Predominantly Express Anti … |
| bulletin of experimental biology and medicine | Quantitative and Qualitative Characterization of Phagocytic Activity of Macrophages of Bone Marrow and Fetal Origin Pubmed: 31183654 |
| Exp Mol Pathol | The regulatory role of SFRP5/WNT5A axis in allergic rhinitis through inhibiting JNK pathway activation and lowering mucin generation in human nasal?¡ 33285209 |
| Vaccine | Co-delivery of PSMA antigen epitope and mGM-CSF with a cholera toxin-like chimeric protein suppressed prostate tumor growth via activating dendritic cells and?¡ 33612342 |
| Catalog No. | Related products for research use of Sus scrofa; Porcine (Pig) Organism species | Applications (RESEARCH USE ONLY!) |
| RPA045Po01 | Recombinant Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) | Positive Control; Immunogen; SDS-PAGE; WB. |
| SEA045Po | ELISA Kit for Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) | Enzyme-linked immunosorbent assay for Antigen Detection. |
| LMA045Po | Multiplex Assay Kit for Colony Stimulating Factor 2, Granulocyte Macrophage (GM-CSF) ,etc. by FLIA (Flow Luminescence Immunoassay) | FLIA Kit for Antigen Detection. |