Packages (Simulation)

Reagent Preparation
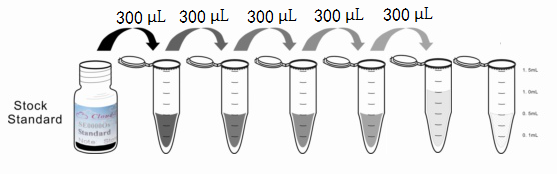
Image (I)
Image (II)
Certificate


Multiplex Assay Kit for Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay)
GOLM1; GP73; C9orf155; Golgi Membrane Protein 1; Golgi Protein 73
(Note: Up to 8-plex in one testing reaction)
- Product No.LMB668Mu
- Organism SpeciesMus musculus (Mouse) Same name, Different species.
- Sample TypeSerum, plasma, tissue homogenates, cell lysates, cell culture supernates and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3.5h
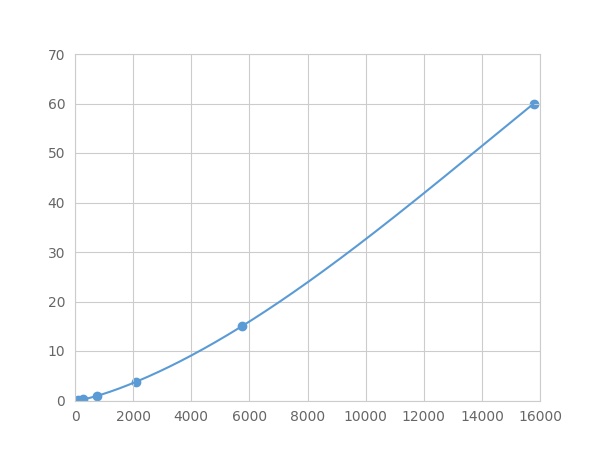
- Detection Range0.06-60ng/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 0.02 ng/mL.
- DownloadInstruction Manual
- UOM 8Plex 7Plex 6Plex 5Plex 4Plex 3Plex 2Plex1Plex
- FOB
US$ 427
US$ 443
US$ 468
US$ 501
US$ 534
US$ 583
US$ 657
US$ 821
Add to Price Calculator
Result
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay).
No significant cross-reactivity or interference between Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay) and the recovery rates were calculated by comparing the measured value to the expected amount of Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 86-94 | 89 |
| EDTA plasma(n=5) | 83-90 | 86 |
| heparin plasma(n=5) | 83-102 | 98 |
| sodium citrate plasma(n=5) | 78-104 | 88 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Golgi Phosphoprotein 2 (GOLPH2) ,etc. by FLIA (Flow Luminescence Immunoassay) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 78-103% | 82-101% | 84-103% | 82-90% |
| EDTA plasma(n=5) | 89-96% | 96-105% | 93-105% | 85-104% |
| heparin plasma(n=5) | 90-98% | 94-102% | 86-96% | 83-91% |
| sodium citrate plasma(n=5) | 90-104% | 87-98% | 85-99% | 79-96% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| 96-well plate | 1 | Plate sealer for 96 wells | 4 |
| Pre-Mixed Standard | 2 | Standard Diluent | 1×20mL |
| Pre-Mixed Magnetic beads (22#:GOLPH2) | 1 | Analysis buffer | 1×20mL |
| Pre-Mixed Detection Reagent A | 1×120μL | Assay Diluent A | 1×12mL |
| Detection Reagent B (PE-SA) | 1×120μL | Assay Diluent B | 1×12mL |
| Sheath Fluid | 1×10mL | Wash Buffer (30 × concentrate) | 1×20mL |
| Instruction manual | 1 |
Assay procedure summary
1. Preparation of standards, reagents and samples before the experiment;
2. Add 100μL standard or sample to each well,
add 10μL magnetic beads, and incubate 90min at 37°C on shaker;
3. Remove liquid on magnetic frame, add 100μL prepared Detection Reagent A. Incubate 60min at 37°C on shaker;
4. Wash plate on magnetic frame for three times;
5. Add 100μL prepared Detection Reagent B, and incubate 30 min at 37°C on shaker;
6. Wash plate on magnetic frame for three times;
7. Add 100μL sheath solution, swirl for 2 minutes, read on the machine.
GIVEAWAYS
INCREMENT SERVICES
| Magazine | Citations |
| Molecular & Cellular Proteomics | LAMC2: A promising new pancreatic cancer biomarker identified by proteomic analysis of pancreatic adenocarcinoma tissues Mcponline: 023507 |
| Afro-Egypt J Infect Endem Dis | Diagnostic and Prognostic Validity of Serum Golgi Protein 73 in Egyptian Patients with Hepatocelluar Carcinoma Ajied: Ajied_System_Files |
| Journal of Applied Pharmaceutical Science | Diagnostic role of Golgi protein 73 and IL-17 in Egyptian cirrhotic rather than hepatocellular carcinoma patients. handle:123456789 |
| Oncology Letters | Serum Golgi protein 73 is a prognostic rather than diagnostic marker in hepatocellular carcinoma pubmed:29113278 |
| Scientific Reports | The possible role of Dickkopf-1, Golgi protein-73 and Midkine as predictors of hepatocarcinogenesis: a review and an Egyptian study Pubmed: 32198440 |
| Hepatobiliary & Pancreatic Diseases International | Dynamic expression of hepatic GP73 mRNA and protein and circulating GP73 during hepatocytes malignant transformation Pubmed: 32171652 |
| Biomolecules | Deoxycholic Acid Upregulates Serum Golgi Protein 73 through Activating NF-¦ÊB Pathway and Destroying Golgi Structure in Liver Disease. Biomolecules 2021, 11?¡ 33540642 |
| ADVANCED RESEARCHES | Search for Effective Serum Tumor Markers for Early Diagnosis of Hepatocellular Carcinoma Associated with Hepatitis C |