Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


ELISA Kit for Lecithin Cholesterol Acyltransferase (LCAT)
phosphatidylcholine-sterol O-acyltransferase; Phospholipid-cholesterol acyltransferase
- Product No.SEJ516Ra
- Organism SpeciesRattus norvegicus (Rat) Same name, Different species.
- Sample TypeSerum, plasma and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3h
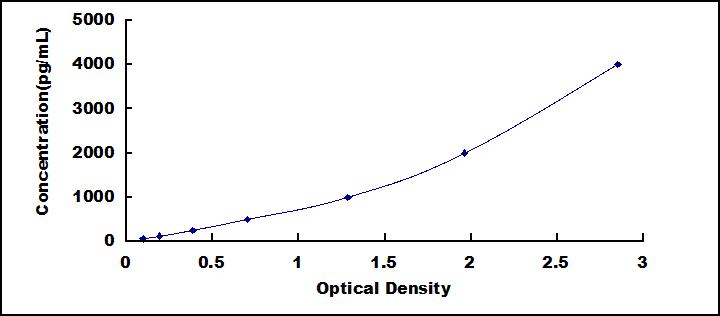
- Detection Range62.5-4,000pg/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 28.4pg/mL.
- DownloadInstruction Manual
- UOM 48T96T 96T*5 96T*10 96T*100
- FOB
US$ 532
US$ 760
US$ 3420
US$ 6460
US$ 53200
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Lecithin Cholesterol Acyltransferase (LCAT).
No significant cross-reactivity or interference between Lecithin Cholesterol Acyltransferase (LCAT) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Lecithin Cholesterol Acyltransferase (LCAT) and the recovery rates were calculated by comparing the measured value to the expected amount of Lecithin Cholesterol Acyltransferase (LCAT) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 90-102 | 97 |
| EDTA plasma(n=5) | 78-96 | 90 |
| heparin plasma(n=5) | 94-102 | 99 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Lecithin Cholesterol Acyltransferase (LCAT) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Lecithin Cholesterol Acyltransferase (LCAT) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Lecithin Cholesterol Acyltransferase (LCAT) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 89-99% | 83-98% | 96-103% | 92-101% |
| EDTA plasma(n=5) | 78-103% | 86-94% | 79-102% | 89-98% |
| heparin plasma(n=5) | 81-102% | 96-105% | 95-105% | 91-98% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.
GIVEAWAYS
INCREMENT SERVICES
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 ELISA Kit Customized Service
ELISA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution -
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
| Magazine | Citations |
| Int J Rheum Dis | Plasma phospholipase, γ‐CEHC and antioxidant capacity in fibromyalgia PubMed: 26585319 |
| Inflammation. | Alteration of Lysophosphatidylcholine-Related Metabolic Parameters in the Plasma of Mice withExperimental Sepsis. pubmed:28028754 |
| British journal of cancer | A combined biomarker panel shows improved sensitivity for the early detection of ovarian cancer allowing the identification of the most aggressive type II tumours. pubmed:28664912 |
| Journal of Agricultural and Food Chemistry | Lipid-lowering effects of medium-chain triglyceride-enriched coconut oil in combination with licorice extracts in experimental hyperlipidemic mice Pubmed: 30244576 |
| Experimental and Therapeutic Medicine | Identification and validation of differentially expressed proteins in serum of CSU patients with different duration of wheals using an iTRAQ labeling, 2D‑LC‑MS/MS Doi: 10.3892/etm.2018.6818 |
| British Journal of Cancer | Diagnosis of epithelial ovarian cancer using a combined protein biomarker panel. Pubmed: 31388184 |
| BIOMEDICINE & PHARMACOTHERAPY | The synergistic mechanism of total saponins and flavonoids in Notoginseng− Safflower pair against myocardial ischemia uncovered by an integrated … Pubmed: 32739736 |
| Biomedicine & Pharmacotherapy | Pharmacokinetics/pharmacometabolomics-pharmacodynamics reveals the synergistic mechanism of a multicomponent herbal formula, Baoyuan decoction?¡ |
| Cell Metab | Defects in a liver-bone axis contribute to hepatic osteodystrophy disease progression Pubmed:35235775 |
| Catalog No. | Related products for research use of Rattus norvegicus (Rat) Organism species | Applications (RESEARCH USE ONLY!) |
| RPJ516Ra02 | Recombinant Lecithin Cholesterol Acyltransferase (LCAT) | Positive Control; Immunogen; SDS-PAGE; WB. |
| RPJ516Ra01 | Recombinant Lecithin Cholesterol Acyltransferase (LCAT) | Positive Control; Immunogen; SDS-PAGE; WB. |
| PAJ516Ra01 | Polyclonal Antibody to Lecithin Cholesterol Acyltransferase (LCAT) | WB; IHC; ICC; IP. |
| PAJ516Ra02 | Polyclonal Antibody to Lecithin Cholesterol Acyltransferase (LCAT) | WB; IHC; ICC; IP. |
| LAJ516Ra71 | Biotin-Linked Polyclonal Antibody to Lecithin Cholesterol Acyltransferase (LCAT) | WB; IHC; ICC. |
| SEJ516Ra | ELISA Kit for Lecithin Cholesterol Acyltransferase (LCAT) | Enzyme-linked immunosorbent assay for Antigen Detection. |
| LMJ516Ra | Multiplex Assay Kit for Lecithin Cholesterol Acyltransferase (LCAT) ,etc. by FLIA (Flow Luminescence Immunoassay) | FLIA Kit for Antigen Detection. |